It’s no secret that honey bee populations have been struggling to survive in recent years. Hypotheses abound in regards to the causes of honey bee decline, and it’s likely that no single factor will prove to be responsible, but it’s clear that the presence of Varroa mites (Varroa destructor) in honey bee hives weakens bees, and contributes significantly to colony demise.

The shiny brown disc attached to the thorax of this honey bee, is the parasitic Varroa mite (click any image to enlarge)
The Varroa mite is an obligate parasite of various species of honey bees. This mite was first introduced into the United States from Asia in 1987. Previously Varroa’s host had been the eastern honey bee (Apis cerana), which was well adapted to this parasite, and seemed to suffer few ill effects from it.

Low temperature scanning electron micrograph (LTSEM) of Varroa destructor on a honey bee host (Image Source: Public Domain - USDA Agricultural Research Service)
Then Varroa found a new host in the European honey bee (Apis mellifera). Unlike the Asian bees, European honey bees were not adapted to living with the mite, and shortly after its introduction to the United States, colonies, both feral and managed, all across the country were devastated. If you’ve read the Aesculus californica post, you might recognize the common theme of co-evolution between these posts. Novel environmental influences, be they parasitic, or toxic, can have detrimental effects on non-adapted populations.

This honey bee has a Varroa mite on her thorax. She was found crawling on the ground outside the hive, ataxic, uncoordinated, and unwilling to fly
Some former beekeepers we’ve spoken to have told us that they had given up beekeeping by the early 1990s because they found it impossible to sustain their bee colonies in the presence of Varroa. Entire apiaries were routinely wiped out. Beekeeping has changed since Varroa’s arrival, and although the situation is improving, many beekeepers still lose their hives due to overwhelming Varroa populations.
The Varroa mite is here to stay. These mites are now found in honey bee colonies worldwide, and they’ve become established on every continent except Australia.[1] Our hives are no exception.
All beekeepers here have Varroa, at least to some extent. Some colonies are showing some resistance to the presence of the mite, but many honey bee colonies are still being significantly, and adversely, affected by Varroa mites.
Varroa’s Life Cycle
In order to understand how any parasite thrives and survives, it’s imperative to understand its life cycle. Varroa mites can only reproduce within the hives of honey bees. Although they’ve occasionally been found on wild native bees, like bumble bees, they can’t survive on wild bee hosts as they depend on the structure of honey bee hives to successively reproduce. They are perfectly adapted to life within a honey bee hive.
Female mites are approximately 1.0 mm long and 1.5 mm wide, and must enter the brood cells where honey bee larva reside before the cells are capped, and have a preference for larger drone brood cells that are capped for a longer period of time.
They feed on the hemolymph of developing bee pupa, and complete their entire life-cycle within the cell before the bees emerge as adults. The following video, although not short, provides an excellent overview of the life-cycle, both of the honey bee, and of the Varroa mite. It may take a minute to load, but if you watch it, you’ll understand how Varroa is able to exploit the honey bee’s development cycle to its advantage.
http://video.google.com/videoplay?docid=-7304562435786960616#
Detecting Varroa in the Hive
As new beekeepers we knew that our colonies would have Varroa, but we needed to learn how to evaluate the overall burden of Varroa within our hives. A number of detection methods are available for assessing mite populations within a colony.
Mites attached to adult bees, called phoretic mites, may periodically be groomed off of the bees, and naturally fall to the hive floor. Monitoring of this natural mite-fall can be used to gauge severity of Varroa infestation in the hive. All of our hives are fitted with IPM screened bottom boards. By using screened bottom boards in the hive, as the mites fall off the bees, most of the mites will fall through the screen onto the ground below. By inserting a monitoring grid (usually called a ‘sticky board’) under the screen, the mites will land on the board, and the rate of mite fall can be monitored for each hive.

Positioning this board under the screen of the hive floor, mites that land on the board can be counted
To accelerate these counts, some beekeepers will motivate grooming in bees by applying a dusting of powdered sugar to bees within the hive. This stimulates grooming, and increases the rate of mite drop. However, we have a lot of ants near the apiary that we’re already struggling to keep at bay, so we avoided the sugar dusting method.
Instead, last week we completed our first passive ‘sticky board’ counts for our colonies. We first labeled each board, as we have multiple hives. Each board was then sprayed with a light even coating of cooking oil.

A food grade spray oil is safe for use under the hive, traps mites on the board, but is easy to clean up
The oil will trap any mites that fall, especially live mites, and prevent them from crawling away before they’re counted. Some advocate the use of conventional orchard sticky-trap sprays, or Vaseline, but we’ve found the advantage of cooking spray is it’s very easily wiped off when you’re done with the mite counts, and doesn’t leave any unwanted residues.

There is a slot under the screen of the screened bottom board that the monitoring board slides into at the bottom of the hive
We installed the oiled boards under each hive for 48 hours. Some beekeepers only leave the boards in for 24 hours, others for 5-7 days. Leaving the boards in slightly longer should give a more accurate measure of natural mite-drop over time. However, it’s advisable not to leave the boards in for too long, especially under large colonies, as more than mites will fall through the screen, complicating the counts.
After 48 hours, each board was removed, and the mites on the board were counted. We determined that two tools help tremendously for doing the counts, especially when you have multiple hives. A magnifying source (we used loupes, but a strong hand lens will work), and a strong light source.
Each board has lettered rows, and numbered columns, making it easy to be methodical while performing counts.
The boards when they’re first removed will have a varied amount of debris, not just mites. Pieces of pollen, and flakes of wax from comb building and remodelling, also fall on the board.

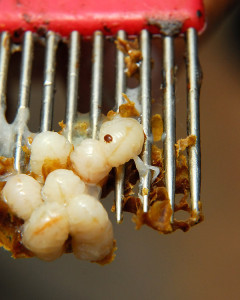
Developmental stage of the mite, orientation of the mite, and amount of debris on the board can all serve to confound getting accurate mite counts. This mite was hiding under some debris...
Unless you have perfect magna-vision, be prepared to spend some time doing the counts. It takes time to train your eye to see the mites amidst the debris field.

Excessive debris may be seen on boards placed under large colonies, or if left in for too many days during the count
We counted the total number of mites for each hive, and then divided that number by 2 to determine our 24 hour mite count.
Chamomile Count: 1; 1/2 = 0.5 mites per 24 hours
Rosemary Count: 8; 8/2 = 4 mites per 24 hours
Lavender Count: 25; 25/2 = 12.5 mites per 24 hours
Salvia Count: 180; 180/2 = 90 mites per 24 hours
The Salvia mite count at first is alarming, and it is too high. Depending on who you talk to, or which papers you read, threshold counts vary, but generally any natural drop count over 60 mites in 24 hours is considered potentially damaging to the colony.
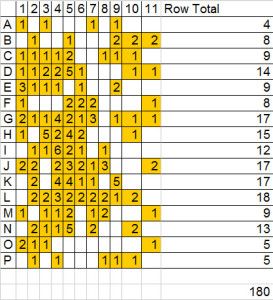
This is how the distribution of mites looked on the sticky board from the Salvia hive (click image to enlarge)
However, natural mite-fall counts are far from perfect. One key factor these counts don’t correct for is the size of the colony. This means we can’t compare the results of Salvia (our largest colony), to Chamomile (our smallest). The difference in population of bees could account for the part of the difference in the number of mites.
A somewhat more quantitative approach for counting mites is to do an alcohol wash. Approximately 300 bees from the brood nest are placed in a jar, and washed down with 250 mls of isopropyl alcohol. The number of mites in the resulting supernatant provides an indication as to the number of total (phoretic) mites, per 300 bees. Note though, this still doesn’t count mites trapped inside the capped cells with the larva/pupa. As such, at some times of year these counts may still woefully underestimate total Varroa burden within the hive. However, as these are mites per (roughly) the same number of adult bees, it is a little easier to compare Varroa load hive to hive.

This alcohol wash was done at a friend's apiary two weeks ago. The Queen was gone, and there was a laying worker in the hive. This failing colony yielded 80+ mites from approximately 300 bees. That translates to a 26.7% infestation rate (a little over 1 in 4 bees)
Despite imperfections with these survey methods, repeat regular counts do help us as beekeepers to gauge when mite populations are increasing considerably, and can help to guide when to intervene, if the beekeeper desires to do so (more on that later).
As we’ve only done a single sticky board count thus far, we’ll repeat these counts every two weeks to gauge how rapidly our mite populations are increasing.
Varroa Vectored Diseases
Varroa mites alone can weaken honey bee colonies, but they also leave them susceptible to numerous opportunistic pathogens. Varroa mites have the capacity to vector specific diseases to bees, including viruses, one of which is now commonly seen in colonies with high mite burdens. Deformed Wing Virus.
Deformed Wing Virus is one of at least 18 different viruses known to afflict the European honey bee. This virus can be passed from Queen to egg, or between bees orally, but is most prevalent in hives strongly associated with high Varroa mite populations. Virus has been detected in significant concentrations within Varroa mites who are capable of vectoring the disease while attached to developing pupa. [2,3,4,5]

Even before our first Varroa counts, we knew something was wrong when we spotted this worker bee crawling on the ground in front of the Salvia hive.
Two weeks ago we found bees crawling on the ground with deformed wings in front of our large swarm colony hive, Salvia. Generally the belief is that survivor stock is more robust than commercially bred bees, but this colony shows that even survivor stock isn’t immune to Varroa infestation. We suspected that although up until this point the colony seemed healthy and robust, that trouble was brewing, and this is what prompted us to begin surveying for Varroa levels in our hives.
Based on our recent observations at the Salvia hive entrance, we would have been surprised if this hive didn’t have a high mite count.
So with the presence of significant numbers of Varroa mites in the Salvia hive, and those bees overtly expressing signs of being infected with Deformed Wing Virus, we now need to decide which methods of Varroa control we’ll employ in our apiary. We’ll discuss our management options, and the controversies surrounding treatment, in a future post…
——————–
[1] Australian Government: Department of Agriculture, Fisheries, and Forestry. Varroa Mite.
[2] Fievet, J; Tentcheva, D; Gauthier, L; De, Miranda, J; Cousserans, F; Colin, Me; Bergoin, M (March 2006). “Localization of deformed wing virus infection in queen and drone Apis mellifera L“. Virology journal 3: 16.
[3] Tentcheva D, Gauthier L, Zappulla N, et al. (December 2004). “Prevalence and seasonal variations of six bee viruses in Apis mellifera L. and Varroa destructor mite populations in France“. Appl. Environ. Microbiol. 70 (12): 7185–91.
[4]Highfield, AC, A El Nagar, L Mackinder, ML Laure, J Noël, MJ Hall, SJ. Martin, and DC. Schroeder (2009) Deformed wing virus implicated in over-wintering honeybee colony losses. Appl. Environ. Microbiol 75(2): 7212-7220.
[5] Runckel, C; Flenniken, M. L.; Engel, J; Ruby, G J; Ganem, D; Andino, R; DeRisi, J. L. (June 2011) Temporal Analysis of the Honey Bee Microbiome Reveals Four Novel Viruses and Seasonal Prevalence of Known Viruses, Nosema, and Crithidia. Plos One Online. Vol 6 Issue 6.













If you want to try formic acid pads, we’ve got more than we need.
Thanks Lisa. We’re considering ordering Thymol strips from Canada (rather than using the gel (Apiguard), or Formic). Is supposed to be a little better tolerated by the bees, and less risky to use during warm weather, albeit not too easy to come by! 😉
Hi CV,
Sorry to hear that there’s something going on in your Salvia hive, hopefully they will manage to overcome the problem and survive.
I watched a show recently and it has an article on the mites in Bees, and a Bee guy (sorry I don’t remember his name) somewhere in the UK has been researching into them and has observed some of his hives excelling in cleaning off the mites. These colonies are still thriving, and by the looks are getting better at it – I guess similar to the Asian Honey Bees…
I hope for the Bee’s sake they adapt quickly and that this behaviour is passed on.
Selecting for ‘hygienic bees’, meaning bees that actually recognize a mite problem, and endeavor to rid themselves of them, is one of the more promising ways of managing varroa. We’ll get more into that in our Varroa Management post. Our bees seem to be removing a number of mite-affected pupa. We could hope that’s enough, but should be prepared in case the mite populations get out of control.
I am sorry your Salvia hive is having problems. The photo of the deformed bee cast from the hive by worker bees is sad. I look forward to reading how you manage the problem. Good luck!
It was quite disheartening to find some of our bees already afflicted with deformed wing virus. This colony has been so large, and so robust, that their rate of reproduction seems to have masked any problems up until now, but it’s now clear that they’re struggling with Varroa. Hopefully, if we can help them manage the mites, the incidence of deformed wing virus will decrease, but we’ll have to wait and see.
Sorry to hear this. Salvia has seemed to be so robust.
Interesting – if disturbing – video. So did I understand it correctly that male mites typically wind up mating with their sisters?
You heard it right Anne, they’re a rather incestuous bunch 😉 This is one time where you’d hope that inbreeding would lead to the demise of a species, but alas, these mites seem rather immune to inbreeding. 🙁
Clare,
Shame to hear this so soon. The organic beekeepers I know use homemade powdered sugar. They don’t like store bought powered sugar because it is mainly corn syrup. So they run cane sugar through the blender to make their powdered sugar.
Two friends came over tonight and we checked out my hive, they were impressed marking 5 weeks. Must have had 20 pounds of capped honey (or syrup). Just a few emerged new bees. No mites or beetles, thought we removed 2 moth larva from under the outside of the hive cover. Next week we are adding another brood box!
Unfortunately, once Varroa is this far advanced, powdered sugar won’t decrease the Varroa population. It can’t get to where the majority of Varroa is, inside the cell. Weekly sugar dusting can help to slow Varroa population growth, so if we’d started in Spring, it may helped, but most recent studies suggest it’s not terribly effective, at least not alone. Early next spring though, we may try it along with drone brood sampling and trapping.
Sounds like you hive is off to a great start! I think our cold weather set all our bees back here, but they are starting to catch up on stores now the weather is warming.
I learn to appreciate the bees in my garden a little bit more after reading this post. I have seen bees crawling and did not look to see what you have described, but will now. Sorry about your Salvia hive.
Having bees has certainly highlighted the variety of issues bees face for us too. We knew bees were in trouble, but until this year hadn’t fully appreciated the wide spectrum of issues they’re facing. Hopefully we can get the Salvia hive straightened out while they still have enough time to build up some good winter reserves. We just have to weigh our management options.
Your posts on the problems affecting bees have been very informative. No wonder they are in decline…they have such a lot to contend with. 🙁
I feel that way too, often. It’s amazing sometimes they survive at all. Our job as beekeepers is to try not to make the situation worse for the bees than it already is.
I wonder why the Salvia bees are more infested with the mites. This is so interesting. I hope you are able to find a way to keep them at bay.
I think I have the answers to that question. The Salvia Queen arrived as a mated Queen. She began laying eggs the first day she arrived (March 30). This was a large swarm to start with, and has produced a tremendous amount of population. By getting off to a strong start in the spring, Varroa has also had a much longer period to reproduce inside this hive, compared to our others. Our afterwarm hive (Lavender) didn’t start laying eggs for almost a month after Salvia (they have the second highest mite population in the apiary). Chamomile and Rosemary were the newest, arriving in May, and had likely been treated previously at the commercial apiary they came from. The higher Varroa count in Rosemary may be due to drift of bees from Salvia, which we’ve suspected for some time based on their increased population. So it’s really all about Varroa having the opportunity to reproduce earlier, and over a longer period. If you plot mite counts in untreated hives, Varroa population growth is usually an exponential curve, so we know we need to address this quickly, as it can get much worse, in a very short span of time.
Given the possible spread of the mites to Rosemary via drift, might you be rethinking the idea of switching Rosemary and Chamomile?
It’s probably more important to get Chamomile moved so they have enough population to build up stores over summer. To swap the hives, we have to do it fairly early in the season though. Once lots of stores are built up, switching the hive can actually set off a serious case of robbing.
Varroa will be in the hives regardless, and will continue to build up in all the hives over the summer and into fall. We’ll just need to continue monitoring to ensure we take any corrective action earlier, rather than later.
This though is why we have more than one hive, and why all our equipment is the same size. Going into winter, we can do some creative swapping of individual frames of food, brood etc. to balance out strong and weak hives. Alternatively, any hives that are deemed too weak to survive winter on their own can be combined into a stronger hive. That’s the problem with beekeeping, too many choices on how to do things!
Like Donna’s comment, each time I read your bee posts I appreciate so much what the bees do in my garden.
For me, as a gardener, it also drives home the critical importance of not polluting the environment of pollinators with chemical sprays. Bees are struggling as it is, and that just makes a bad situation that much worse. It also motivates me to plant a much more diverse array of nectar and pollen producing plants in the garden too, to help ensure there’s always something available for the bees, and all of our other pollinators too! Every little bit helps, and with very little effort every gardener can have a positive impact on their local pollinator populations.
Great post! My new hive has small-cell comb because my stepfather took a class that promoted it as a varroa deterrent. I don’t mind having the small-cell comb, but when I got online to research it I found articles like this that left me skeptical:
http://www.beebehavior.com/small_cell_comb_varroa_mites.php
I really appreciate your detailed diagrams of your mite count. I wonder if the data would be skewed for a hive as new as mine (since the bee population is low).
In a recent beekeeping class we reviewed a lot of the recent scientific literature in regards to parasites and diseases of bees, and it seems that the small-cell study that originally advocated using small cell comb to limit Varroa was very poorly designed and executed. A number of studies since then have debunked the small-cell/Varroa myth.
Once I fully understood the Varroa lifecycle, what became obvious was that in order to limit Varroa’s population growth, you have to employ management techniques early, even if all you’re doing is dusting with powdered sugar and drone trapping, the earlier the better. We should have started the day the bees were hived. If we’d sugar-dusted the first swarm as we hived them, we may have knocked the Varroa population down significantly enough early on that numbers would not be as high now. Of course, there’s no one ideal control method, but there are a number of options available, and in our next Varroa post I explain how we’re going to try to manage it (of course it will change as it needs too if what we do doesn’t work). 😉
As for counts, I’d start now. The first count will only give you a baseline reading for each hive. The counts after that, how ever often you choose to do them, should give you an idea as to how quickly Varroa is reproducing within the hive.
You seem to be very calm about this…
I’m not sure if I’m calm, or just resigned to the fact that Varroa is a very real part of hive life these days. ;)That said though, we do need to address the level of mites in the Salvia hive as soon as possible. At the moment, the only thing going in their favor is that this colony started out very strong. Hopefully we can get the mite levels down soon, so this colony can focus on building their winter stores.
You got me in gear to do our own varroa inspection. Our 24 hour count is 13 and that’s with using the powdered sugar method.
That’s more in line with our other hives, and a pretty good count. Sure beats 180! If you continue to dust weekly, and/or employ drone trapping, hopefully you can keep the count that low through August. Any other swarms we catch are going to be dusted on arrival, and the drone frames go in then too! 😛